 (a) Relative energies of subshells at the time they are being filled. Thus, the electron configuration of cobalt is 1s2 2s 2 2p6 3s2 3p6 4s2 3d7. History and Uses: Cobalt was discovered by Georg Brandt, a Swedish chemist, in 1739. Seven electrons get filled to 3d orbitals. Electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d7. The six electrons and two electrons enter 3p and 4s respectively. In cobalt’s electron configuration, the innermost shell is the 1s orbital, which contains 2 electrons.The second shell consists of the 2s and 2p orbitals, which are completely filled with 2 and 6 electrons, respectively.The third shell includes the 3s and 3p orbitals, which are also fully occupied with 2 and 6 electrons, respectively. 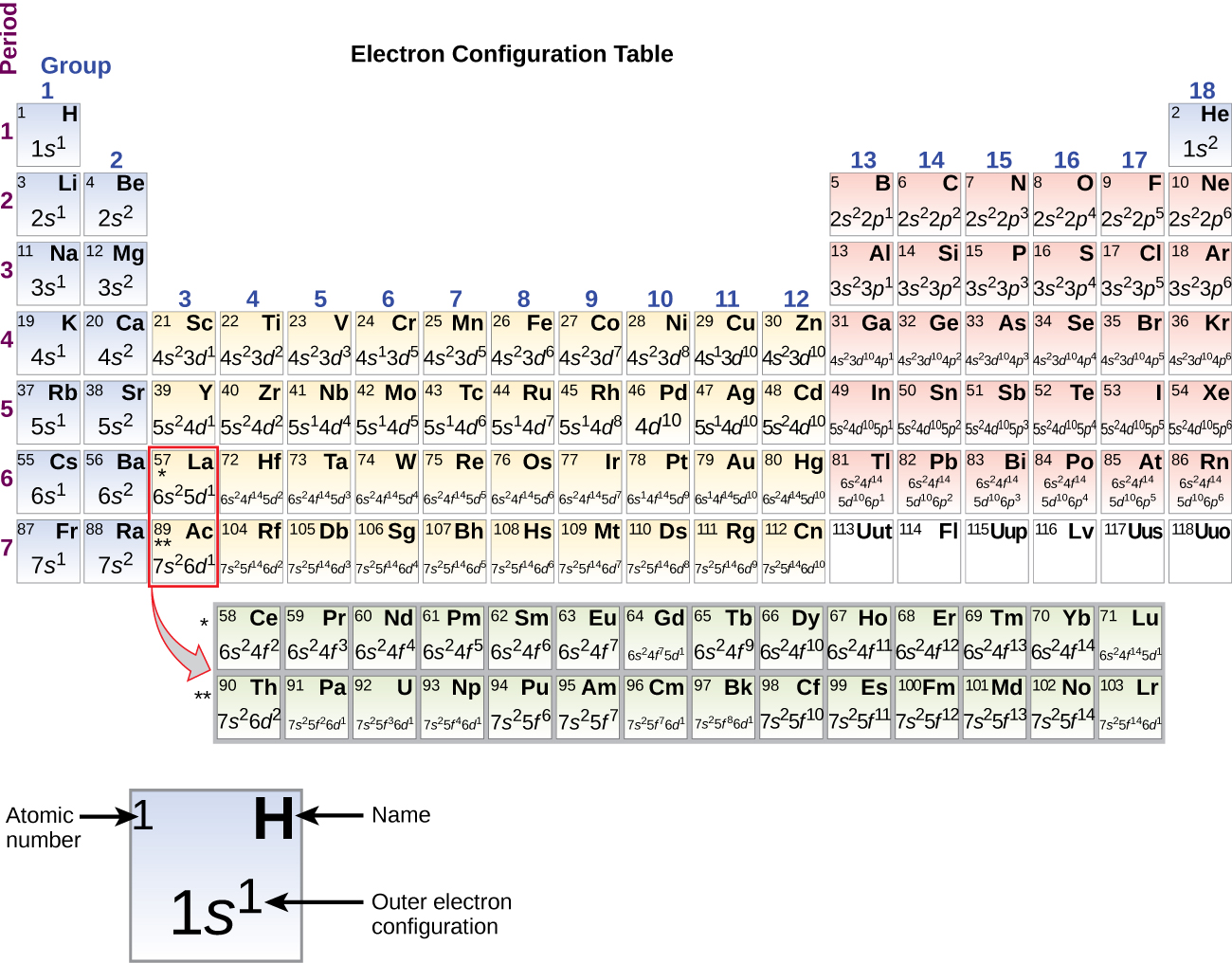 
\) can both react with water, but K has a more radical reaction because it has more shells of core electrons which makes the valence electron in its outermost orbital much easier to lose than the valence electron of Na. 1 Order of filling subshells in the building-up of atomic electron configurations. The first four electrons of cobalt get filled in the 1s and 2s orbitals respectively in the increasing energy level. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
